Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2
You know the right answer?
BEING TIMED. PLS HELP. WILL MARK
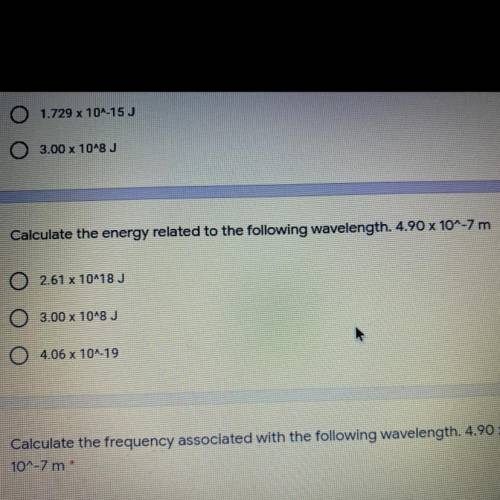
Calculate the energy related to the following wavelength. 4.90 x 1...
Questions


English, 25.06.2019 10:30

Health, 25.06.2019 10:30



History, 25.06.2019 10:30





Mathematics, 25.06.2019 10:30


History, 25.06.2019 10:30



Mathematics, 25.06.2019 10:30

Mathematics, 25.06.2019 10:30

History, 25.06.2019 10:30

Mathematics, 25.06.2019 10:30