Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:20
Amixture of gaseous sulfur dioxide and oxygen are added to a reaction vessel and heated to 1000 k where they react to form so3(g). if the vessel contains 0.669 atm so2(g), 0.395 atm o2(g), and 0.0851 atm so3(g) after the system has reached equilibrium, what is the equilibrium constant kp for the reaction: 2 so2(g) o2(g) ⇌ 2 so3(g)
Answers: 3

Chemistry, 21.06.2019 23:00
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1


Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1
You know the right answer?
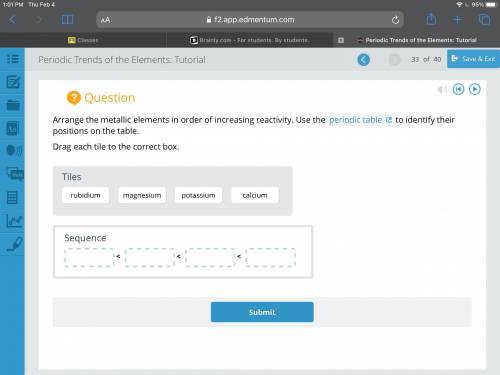
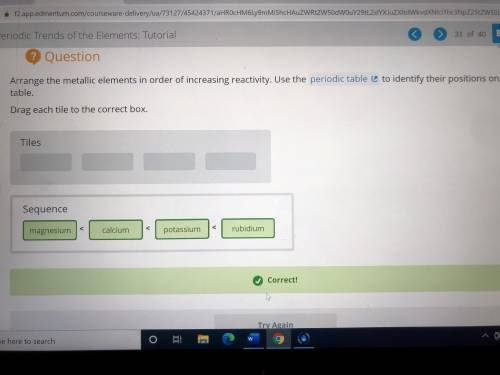
Arrange the metallic elements in order of increasing reactivity. Use the periodic table to identify...
Questions


Advanced Placement (AP), 01.12.2020 17:40

Mathematics, 01.12.2020 17:40


English, 01.12.2020 17:40





Mathematics, 01.12.2020 17:40

Mathematics, 01.12.2020 17:40

English, 01.12.2020 17:40




Mathematics, 01.12.2020 17:40

Business, 01.12.2020 17:40

Mathematics, 01.12.2020 17:40

Mathematics, 01.12.2020 17:40

Mathematics, 01.12.2020 17:40