Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2


Chemistry, 22.06.2019 23:00
If two identical atoms are bonded,what kind of molecule is formed
Answers: 1
You know the right answer?
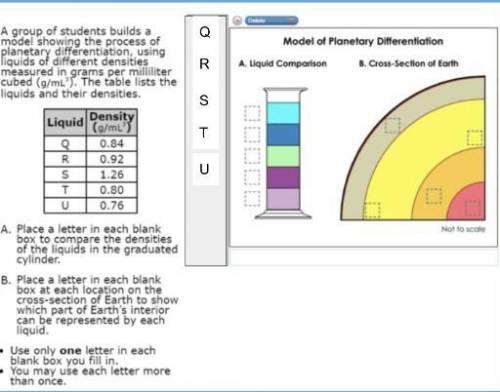
A group of students build a model showing the process of planetary differentiation using liquids of...
Questions


English, 05.05.2021 07:40

Mathematics, 05.05.2021 07:40

Mathematics, 05.05.2021 07:40



English, 05.05.2021 07:50


Mathematics, 05.05.2021 07:50

Chemistry, 05.05.2021 07:50

History, 05.05.2021 07:50

World Languages, 05.05.2021 07:50


English, 05.05.2021 07:50

Chemistry, 05.05.2021 07:50


Chemistry, 05.05.2021 07:50



History, 05.05.2021 07:50