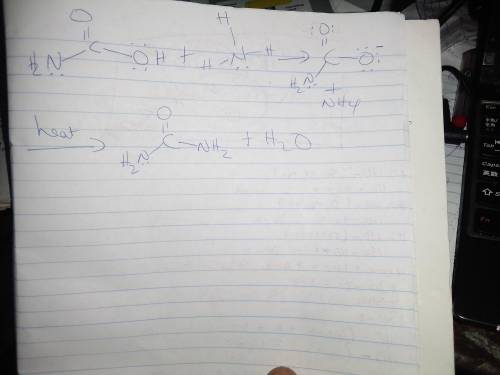Chemistry, 05.02.2021 22:00 saucyboyFredo
Here is the reaction of carbamic acid and ammonia to form an amide and water. There is a scheme of a reversible reaction where carbamic acid reacts with ammonia to give water and an unknown compound. Carbamic acid is H2NCOH with an oxygen atom attached to the carbon atom by a double bond. Ammonia is a nitrogen atom with three H atoms attached. Water is HOH. Draw the amide product of this reaction.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Ahypothesis must be testable and falsifiable to be considered scientific a. trueb. false
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 23.06.2019 05:00
How many atomic mass units are equal to 1.672×10−24 g of protons?
Answers: 3

Chemistry, 23.06.2019 11:00
Nh4no3 n2o + 2h2o a chemist who is performing this reaction starts with 160.1 g of nh4no3. the molar mass of nh4no3 is 80.03 g/mol; the molar mass of water (h2o) is 18.01 g/mol. what mass, in grams, of h2o is produced?
Answers: 1
You know the right answer?
Here is the reaction of carbamic acid and ammonia to form an amide and water. There is a scheme of a...
Questions

English, 20.11.2019 23:31


History, 20.11.2019 23:31

English, 20.11.2019 23:31

English, 20.11.2019 23:31

Mathematics, 20.11.2019 23:31

Geography, 20.11.2019 23:31

English, 20.11.2019 23:31

Mathematics, 20.11.2019 23:31





Chemistry, 20.11.2019 23:31

Mathematics, 20.11.2019 23:31


Arts, 20.11.2019 23:31


Biology, 20.11.2019 23:31

Social Studies, 20.11.2019 23:31