
Chemistry, 05.02.2021 22:00 catsareokiguess
A solid sample of Zinc Hydroxide is added to 0.350 L of 0.500 M aqueous Hydrogen Bromide. The solution that remains is still acidic. It is then titrated with 0.500 M NaOH solution, and it takes 88.5 mL of the NaOH solution to reach the equivalence point. What mass of Zinc Hydroxide was added to the Hydrogen Bromide solution?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2
You know the right answer?
A solid sample of Zinc Hydroxide is added to 0.350 L of 0.500 M aqueous Hydrogen Bromide. The soluti...
Questions

Mathematics, 07.09.2021 18:00


Mathematics, 07.09.2021 18:00


Biology, 07.09.2021 18:00

Medicine, 07.09.2021 18:00





Mathematics, 07.09.2021 18:00

History, 07.09.2021 18:00

Chemistry, 07.09.2021 18:00


Mathematics, 07.09.2021 18:00

Mathematics, 07.09.2021 18:00

Mathematics, 07.09.2021 18:00


Mathematics, 07.09.2021 18:00

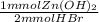 = 65.375 mmol Zn(OH)₂.
= 65.375 mmol Zn(OH)₂.


