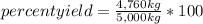A chemical process used to produce ethanol as a fuel additive was expected to produce 5,000 kilograms of ethanol based on the amounts of starting materials used, but only 4,760 kilograms were produced. What was the percent yield for ethanol in this process? A)1.09 percent B)4.80 percent C)95.2 percent D)105 percent

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3
You know the right answer?
A chemical process used to produce ethanol as a fuel additive was expected to produce 5,000 kilogram...
Questions

History, 21.11.2019 06:31


Mathematics, 21.11.2019 06:31

Health, 21.11.2019 06:31

Mathematics, 21.11.2019 06:31


History, 21.11.2019 06:31

Social Studies, 21.11.2019 06:31

Chemistry, 21.11.2019 06:31



English, 21.11.2019 06:31




History, 21.11.2019 06:31

History, 21.11.2019 06:31

Mathematics, 21.11.2019 06:31