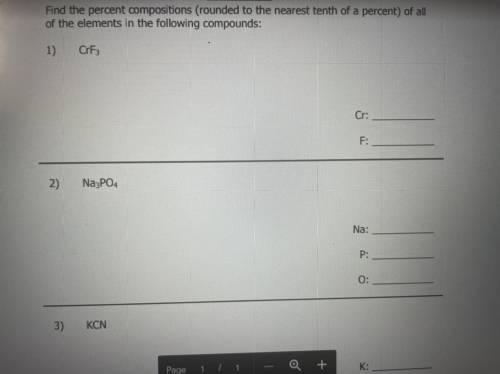
Who can help me with the process and the answer?
...

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Strong conductivity of plasma allows it to act and react as and
Answers: 2

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

You know the right answer?
Questions



Biology, 16.12.2020 08:00

Mathematics, 16.12.2020 08:00


Mathematics, 16.12.2020 08:00

Biology, 16.12.2020 08:00

Mathematics, 16.12.2020 08:00


Mathematics, 16.12.2020 08:00

Mathematics, 16.12.2020 08:00

Geography, 16.12.2020 08:00


History, 16.12.2020 08:00

Mathematics, 16.12.2020 08:00


Biology, 16.12.2020 08:00


Physics, 16.12.2020 08:00

Mathematics, 16.12.2020 08:00