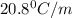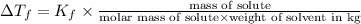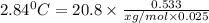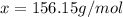Chemistry, 08.02.2021 06:50 patricklcc07777
Menthol is a crystalline substance with a peppermint taste and odor. When 0.533 g of menthol is dissolved in 25.0 g of cyclohexane, the freezing point of the solution is lowered by 2.84 ∘C. Look up the freezing point and f constant for cyclohexane in the Colligative Constants table. Calculate the molar mass of menthol.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which of the following pairs of elements belong to the same groupa. h and he b. li and bec. c and pb d. ga and ge
Answers: 1

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2
You know the right answer?
Menthol is a crystalline substance with a peppermint taste and odor. When 0.533 g of menthol is diss...
Questions




Biology, 15.07.2019 15:00



Biology, 15.07.2019 15:00


Biology, 15.07.2019 15:00





Mathematics, 15.07.2019 15:00

Physics, 15.07.2019 15:00


Social Studies, 15.07.2019 15:00

Social Studies, 15.07.2019 15:00

Social Studies, 15.07.2019 15:00

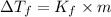
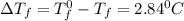 = Depression in freezing point
= Depression in freezing point
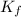 = freezing point constant =
= freezing point constant =