
Chemistry, 09.02.2021 01:50 courtney3652
The equilibrium constant, Kc, for the following reaction is 83.3 at 500 K. PCl3(g) Cl2(g) PCl5(g) Calculate the equilibrium concentrations of reactant and products when 0.453 moles of PCl3 and 0.453 moles of Cl2 are introduced into a 1.00 L vessel at 500 K.

Answers: 1
Another question on Chemistry

Chemistry, 20.06.2019 18:04
4moles of nitrogen gas are confined to a 6.0 l vessel at 177 °c and 12.0 atm. if the vessel is allowed to expand isothermally to 36.0 l, what would be the final pressure?
Answers: 3

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2

Chemistry, 23.06.2019 02:30
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
You know the right answer?
The equilibrium constant, Kc, for the following reaction is 83.3 at 500 K. PCl3(g) Cl2(g) PCl5(g) Ca...
Questions

History, 27.10.2019 17:43

Chemistry, 27.10.2019 17:43


Mathematics, 27.10.2019 17:43



Mathematics, 27.10.2019 17:43


Mathematics, 27.10.2019 17:43



Mathematics, 27.10.2019 17:43

Arts, 27.10.2019 17:43

World Languages, 27.10.2019 17:43




Health, 27.10.2019 17:43

Chemistry, 27.10.2019 17:43

Mathematics, 27.10.2019 17:43

![[PCl_3]=[Cl_2]=0.068M](/tpl/images/1103/3296/1ad55.png)
![[PCl_5]=0.385M](/tpl/images/1103/3296/9d9e6.png)
![Kc=\frac{[PCl_5]}{[Cl_2][PCl_3]}](/tpl/images/1103/3296/6b2bc.png)
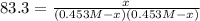
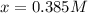
![[PCl_3]=[Cl_2]=0.453M-0.385M=0.068M](/tpl/images/1103/3296/82c25.png)


