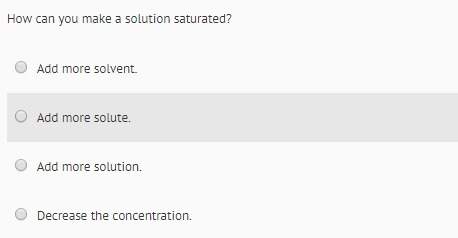
Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1
You know the right answer?
Hot lead with a mass of 200.0 g of (Specific heat of Pb = 0.129 J/g˚C) at 176.4˚C was dropped into a...
Questions

English, 11.12.2021 22:40


Mathematics, 11.12.2021 22:40



Mathematics, 11.12.2021 22:40



Mathematics, 11.12.2021 22:40

Business, 11.12.2021 22:40


English, 11.12.2021 22:40

History, 11.12.2021 22:40

Mathematics, 11.12.2021 22:40

Mathematics, 11.12.2021 22:40


Mathematics, 11.12.2021 22:40



Health, 11.12.2021 22:40