
Chemistry, 11.02.2021 01:40 hihudgins902
The reaction heated up 100 g of water in the beaker from 25 °C to 50.0 °C. The specific heat capacity of water is 4.184 J/g°C. Calculate the amount of heat energy that was released by the reaction.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 06:30
What is the correct term for living the most sustainable life you can within your current circumstances?
Answers: 1

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2
You know the right answer?
The reaction heated up 100 g of water in the beaker from 25 °C to 50.0 °C. The specific heat capacit...
Questions


Biology, 09.03.2022 22:40


Social Studies, 09.03.2022 22:40


Mathematics, 09.03.2022 22:40


English, 09.03.2022 22:40



Mathematics, 09.03.2022 22:40

History, 09.03.2022 22:40


SAT, 09.03.2022 22:40

Mathematics, 09.03.2022 22:50

Mathematics, 09.03.2022 22:50

Geography, 09.03.2022 22:50

Mathematics, 09.03.2022 22:50


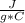 m= 100 gΔT= Tfinal - Tinitial= 50 C - 25 C= 25 C
m= 100 gΔT= Tfinal - Tinitial= 50 C - 25 C= 25 C

