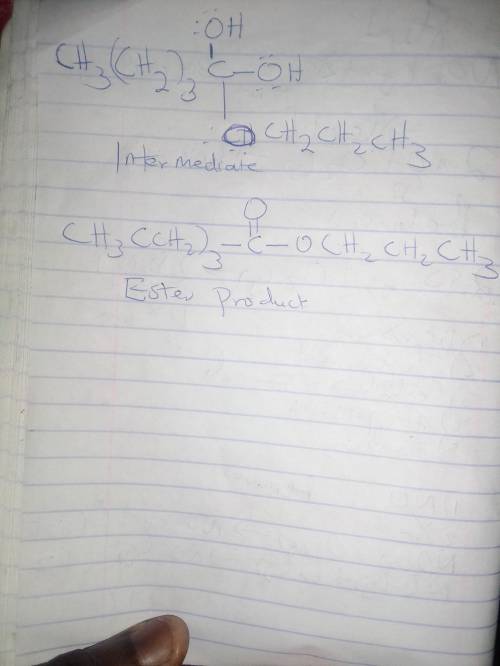Chemistry, 11.02.2021 18:00 unknown1246
In the Fischer esterification reaction, a carboxylic acid reacts with an excess of alcohol in acidic conditions to form an ester. During the reaction the sp2sp2 hybridized carbonyl carbon of the acid forms an sp3sp3 hybridized intermediate before returning to sp2sp2 hybridization in the product. Draw the structure of the neutral sp3sp3 hybridized intermediate and the ester product in the reaction between pentanoic acid and n‑propanol.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:10
How is 0.00235 expressed in proper scientific notation? a. 2.35 × 10-3 b. 0.235 × 10-2 c. 2.35 d. 2.35 × 103
Answers: 1

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

You know the right answer?
In the Fischer esterification reaction, a carboxylic acid reacts with an excess of alcohol in acidic...
Questions




Biology, 24.08.2021 02:50



Mathematics, 24.08.2021 02:50


English, 24.08.2021 02:50



Mathematics, 24.08.2021 02:50

English, 24.08.2021 02:50

Social Studies, 24.08.2021 02:50

Mathematics, 24.08.2021 02:50