Chemistry, 11.02.2021 22:20 angelinagiraffp538zb
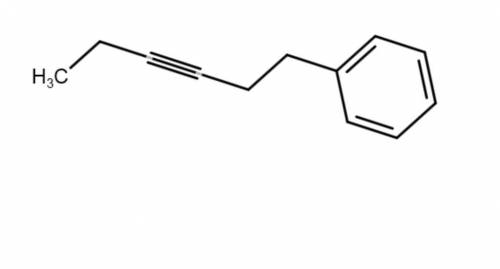
This molecule can be synthesized from an alkyne anion and an alkyl bromide. However, there are two ways in which this molecule can be formed. One way uses a higher molecular weight alkyne anion (Part 1) and the other uses a lower molecular weight anion (Part 2). Draw the two versions in the boxes below. Omit spectator ions.
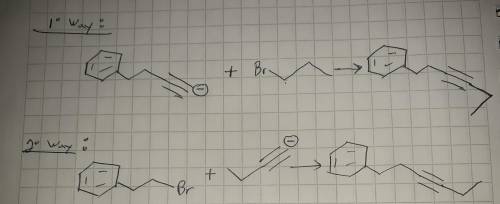
For Part 1: Draw the reactants (i. e., alkyne anion and alkyl bromide) needed for the pathway that uses a higher molecular weight alkyne anion:
For Part 2: Draw the reactants (i. e., alkyne anion and alkyl bromide) needed for the pathway that uses a lower molecular weight alkyne anion:

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5 m hcl? show all of the work needed to solve this problem. mg (s) + 2hcl (aq) → mgcl2 (aq) + h2 (g)
Answers: 3

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2


Chemistry, 23.06.2019 09:30
The earth's surface is (science) a: studied using seismic waves b: constantly changing over time c: only studied indirectly d: the same today as million of years
Answers: 1
You know the right answer?
This molecule can be synthesized from an alkyne anion and an alkyl bromide. However, there are two w...
Questions

Physics, 06.07.2019 18:30

Physics, 06.07.2019 18:30

Mathematics, 06.07.2019 18:30

Biology, 06.07.2019 18:30





History, 06.07.2019 18:30



Physics, 06.07.2019 18:30


Biology, 06.07.2019 18:30


Health, 06.07.2019 18:30

Chemistry, 06.07.2019 18:30

English, 06.07.2019 18:30

History, 06.07.2019 18:30

Chemistry, 06.07.2019 18:30