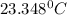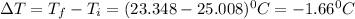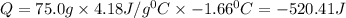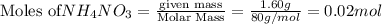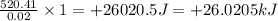Chemistry, 12.02.2021 01:40 pgfrkypory2107
In a coffee cup calorimeter, 1.60 g of NH4NO3 is mixed with 75.0 g of water at an initial temperature of 25.008C. After dissolution of the salt, the final temperature of the calorimeter contents is 23.348C. Assuming the solution has a heat capacity of 4.18 J 8C21 g21 and assuming no heat loss to the calorimeter, calculate the enthalpy change for the dissolution of NH4NO3 in units of kJ/mol.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:40
If equal masses of the listed metals were collected , which would have a greatest volume ? a. aluminum 2.70,b.zinc7.14,c.copper 8.92,d.lead 11.34
Answers: 2

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 05:30
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2
You know the right answer?
In a coffee cup calorimeter, 1.60 g of NH4NO3 is mixed with 75.0 g of water at an initial temperatur...
Questions



Mathematics, 27.08.2021 18:40


Mathematics, 27.08.2021 18:40

English, 27.08.2021 18:40

Social Studies, 27.08.2021 18:40

Chemistry, 27.08.2021 18:40

English, 27.08.2021 18:40


Mathematics, 27.08.2021 18:40






Social Studies, 27.08.2021 18:40


Physics, 27.08.2021 18:40

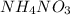 is +26.0205 kJ/mol
is +26.0205 kJ/mol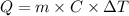
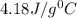
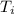 =
= 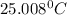
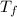 =
=