
Chemistry, 12.02.2021 07:20 winterblackburn78
In ideal gas equation calculations, expressing pressure in Pascals (Pa), necessitates the use of the gas constant, R, equal to . In ideal gas equation calculations, expressing pressure in Pascals (Pa), necessitates the use of the gas constant, R, equal to . 8.314 m3-Pa/mol-K 0.08206 atm L mol-1K-1 1.987 cal mol-1K-1 62.36 L torr mol-1K-1 none of the above

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
You know the right answer?
In ideal gas equation calculations, expressing pressure in Pascals (Pa), necessitates the use of the...
Questions


Mathematics, 11.03.2020 03:03


Biology, 11.03.2020 03:03


History, 11.03.2020 03:03











Computers and Technology, 11.03.2020 03:03



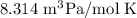
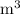
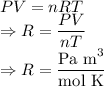
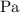 is used so the gas constant value that must be used is
is used so the gas constant value that must be used is 

