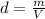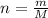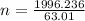Chemistry, 14.02.2021 14:00 patrick171888
A nitric acid solution containing 71.0% HNO3 (by mass) is available in a laboratory. How many moles of HNO3 are present in 1.98 L of this solution?
a.
62.8 mol
b.
31.7 mol
c.
22.3 mol
d.
0.0316 mol
e.
none of these

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which piece of equipment would me most useful for measuring the volume of some water? a. pan balance b. graduated cylinder c. tweezers d. flask quick
Answers: 2


Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1

Chemistry, 23.06.2019 00:50
50 points! need answer asap. what type of organic compound contains the following functional group? (2 points)
Answers: 3
You know the right answer?
A nitric acid solution containing 71.0% HNO3 (by mass) is available in a laboratory. How many moles...
Questions

Physics, 18.03.2021 02:00

Physics, 18.03.2021 02:00

Mathematics, 18.03.2021 02:00




Mathematics, 18.03.2021 02:00




Mathematics, 18.03.2021 02:00

Biology, 18.03.2021 02:00



English, 18.03.2021 02:00


Mathematics, 18.03.2021 02:00


Physics, 18.03.2021 02:00