
Chemistry, 15.02.2021 04:30 pleasehelp5334me2
Hey, can someone help me with this :( I've looked all over my book and I can't figure it out. it's a true or false question and I have a limited amount of tries
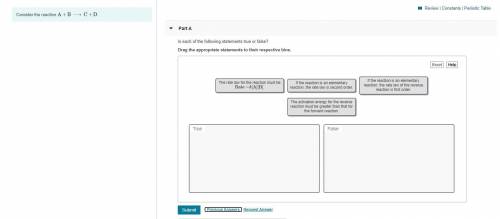

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3
You know the right answer?
Hey, can someone help me with this :( I've looked all over my book and I can't figure it out. it's a...
Questions

Mathematics, 27.06.2019 21:00


Mathematics, 27.06.2019 21:00


Mathematics, 27.06.2019 21:00

English, 27.06.2019 21:00

English, 27.06.2019 21:00

Biology, 27.06.2019 21:00



English, 27.06.2019 21:00

Social Studies, 27.06.2019 21:00





Mathematics, 27.06.2019 21:00





