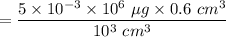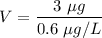Chemistry, 15.02.2021 20:30 fluffylove83
A variation of the indicator-dilution method (see preceding problem) is used to measure total blood volume. A known amount of a tracer is injected into the bloodstream and disperses uniformly throughout the circulatory system. A blood sample is then withdrawn, the tracer concentration in the sample is measured, and the measured concentration [which equals (tracer injected)/(total blood volume) if no tracer is lost through blood vessel walls] is used to determine the total blood volume. In one such experiment, 0.60 \mathrm{cm}^{3}0.60cm 3 of a solution containing 5.00 mg/L of a dye is injected into an artery of a grown man. About 10 minutes later, after the tracer has had time to distribute itself uniformly throughout the bloodstream, a blood sample is withdrawn and placed in the sample chamber of a spectrophotometer. A beam of light passes through the chamber, and the spectrophotometer measure the intensity of the transmitted beam and displays the value of the solution absorbance (a quantity that increases with the amount of light absorbed by the sample). The value displayed is 0.18. A calibration curve of absorbance A versus tracer concentration C (micrograms dye/liter blood) is a straight line through the origin and the point (A=0.9, C=3 \mu \mathrm{g} / \mathrm{L})(A=0.9,C=3μg/L). Estimate the patient's total blood volume from these data.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.
Answers: 3

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 06:30
Suppose a lab group reports a ppercent yield of sand of 105. is it really possible to collect more sand than was originally represented? what is the possible explanation for the extra product?
Answers: 2

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2
You know the right answer?
A variation of the indicator-dilution method (see preceding problem) is used to measure total blood...
Questions



Computers and Technology, 24.08.2019 15:20

Mathematics, 24.08.2019 15:20




SAT, 24.08.2019 15:20

Physics, 24.08.2019 15:20




Computers and Technology, 24.08.2019 15:20



Mathematics, 24.08.2019 15:20




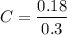
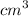 and 5 μg/L was injected.
and 5 μg/L was injected.