
. Carbamide (CH4N2O), can be synthesized by the reaction of ammonia (NH3) with carbon
dioxide (CO2):
2 NH3 (aq) + CO2 (aq) CH4N2O (aq) + H2O (l)
An industrial synthesis of urea obtains 87.5 kg of urea upon reaction of 68.2 kg of ammonia
with 105 kg of carbon dioxide. Determine the limiting reactant, theoretical yield of urea and percent yeild for the reaction

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2

Chemistry, 23.06.2019 13:00
Which of the following statements is true about both nuclear fusion and nuclear fission? they occur in the sun. heavy atoms are split. two light nuclei combine. some mass changes into energy.
Answers: 1
You know the right answer?
. Carbamide (CH4N2O), can be synthesized by the reaction of ammonia (NH3) with carbon
dioxide (CO2)...
Questions

Mathematics, 08.12.2020 04:00

Mathematics, 08.12.2020 04:00

Computers and Technology, 08.12.2020 04:00


Mathematics, 08.12.2020 04:00


Mathematics, 08.12.2020 04:00


Mathematics, 08.12.2020 04:00

Mathematics, 08.12.2020 04:00


Mathematics, 08.12.2020 04:00

History, 08.12.2020 04:00



Mathematics, 08.12.2020 04:00


Chemistry, 08.12.2020 04:00

History, 08.12.2020 04:00

History, 08.12.2020 04:00

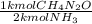 = 2.00 kmol CH₄N₂O
= 2.00 kmol CH₄N₂O

