Chemistry, 19.02.2021 01:00 urstruulyemily
An analytical chemist is titrating of a solution of ethylamine with a solution of . The of ethylamine is . Calculate the pH of the base solution after the chemist has added of the solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added. g

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1
You know the right answer?
An analytical chemist is titrating of a solution of ethylamine with a solution of . The of ethylamin...
Questions

History, 30.10.2020 21:00





Arts, 30.10.2020 21:00



Mathematics, 30.10.2020 21:00

Mathematics, 30.10.2020 21:00


Mathematics, 30.10.2020 21:00

Chemistry, 30.10.2020 21:00


Health, 30.10.2020 21:00

Mathematics, 30.10.2020 21:00

Mathematics, 30.10.2020 21:00

Mathematics, 30.10.2020 21:00


Computers and Technology, 30.10.2020 21:00

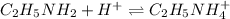
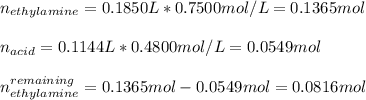
![[ethylamine]=\frac{0.0816mol}{0.1850L+0.1144L}=0.2725M](/tpl/images/1129/1639/9c401.png)
![[salt]=\frac{0.0549mol}{0.1850L+0.1144L}=0.1834M](/tpl/images/1129/1639/c810c.png)
![pOH=pKb+log(\frac{[salt]}{[base]} )\\\\pOH=3.19+log(\frac{0.1834M}{0.2725M})\\\\pOH=3.0](/tpl/images/1129/1639/77c7e.png)