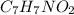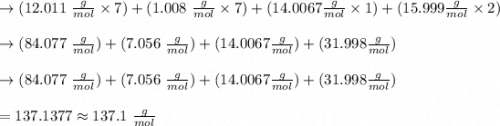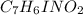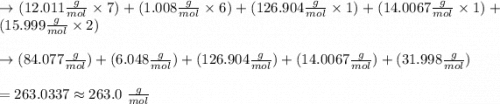onsider the iodination of salicylamide by sodium iodide and sodium hypochlorite via an electrophilic aromatic substitution to form iodo-salicylamide. Reaction scheme illustrating the iodination of salicylamide by sodium iodide and sodium hypochlorite via an electrophilic aromatic substitution to form iodo-salicylamide. Calculate the molar masses of the reactants and product. Report molar masses to 1 decimal place.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which traits do human embryos have that link them to a common ancestor with fish and reptiles? a. scales and tail b. gill slits and scales c. tail and gill slits d. hair and tail
Answers: 2

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

You know the right answer?
onsider the iodination of salicylamide by sodium iodide and sodium hypochlorite via an electrophilic...
Questions

English, 28.08.2019 01:10

Mathematics, 28.08.2019 01:10


Physics, 28.08.2019 01:10



Biology, 28.08.2019 01:10


Mathematics, 28.08.2019 01:10



Mathematics, 28.08.2019 01:10


Social Studies, 28.08.2019 01:10


English, 28.08.2019 01:10




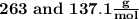 ".
".