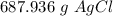Chemistry, 20.02.2021 05:00 Hcalhoun21
How many grams of silver chloride can be produced by reacting excess silver nitrate with 2.4 moles of zinc chloride? AgNO3 + ZnCl2 AgCl + Zn(NO3)2

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1


Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3
You know the right answer?
How many grams of silver chloride can be produced by reacting excess silver nitrate with 2.4 moles o...
Questions

Mathematics, 18.04.2020 06:00


Mathematics, 18.04.2020 06:00

German, 18.04.2020 06:00

Chemistry, 18.04.2020 06:00

Mathematics, 18.04.2020 06:00

History, 18.04.2020 06:00


Mathematics, 18.04.2020 06:00

Mathematics, 18.04.2020 06:00

Spanish, 18.04.2020 06:00



Mathematics, 18.04.2020 06:00

Biology, 18.04.2020 06:00


Mathematics, 18.04.2020 06:02


Chemistry, 18.04.2020 06:16

History, 18.04.2020 06:16

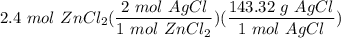 [DA] Multiply/Divide [Cancel out units]:
[DA] Multiply/Divide [Cancel out units]: