
Chemistry, 22.02.2021 18:50 Svetakotok
A 52.9g sample of brass, which has a specific heat capacity of 0.375·J·g^−1°C^−1, is put into a calorimeter (see sketch at right) that contains 100.0g of water. The temperature of the water starts off at 15.0°C. When the temperature of the water stops changing it's 18.4°C. The pressure remains constant at 1 atm. Calculate the initial temperature of the brass sample.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2


Chemistry, 23.06.2019 14:00
[07.06] which of the following chemical reactions is an oxidation-reduction reaction? (2 points) wo3 + 3h2 yields w + 3h2o kno3 + licl yields lino3 + kcl caso4 + 2nacl yields na2so4 + cacl2 mg(no3)2 + 2hbr yields mgbr2 + 2hno3
Answers: 1
You know the right answer?
A 52.9g sample of brass, which has a specific heat capacity of 0.375·J·g^−1°C^−1, is put into...
Questions





Mathematics, 10.01.2020 00:31


Mathematics, 10.01.2020 00:31


History, 10.01.2020 00:31


Biology, 10.01.2020 00:31

Mathematics, 10.01.2020 00:31


History, 10.01.2020 00:31


Mathematics, 10.01.2020 00:31


History, 10.01.2020 00:31


Mathematics, 10.01.2020 00:31

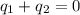
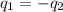
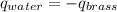
![m_{w}.c_{w}.\Delta T=-[m_{b}.c_{b}.\Delta T]](/tpl/images/1136/4104/e97cd.png)
![100.4.18.(18.4-15)=-[52.9.0.375.(18.4-T)]](/tpl/images/1136/4104/69ce0.png)


