
Chemistry, 30.09.2019 03:30 ConfusedJuliana
a solution was prepared by dissolving 177 mg of potassium sulfate (k2so4, mw = 174.24 g/mol) in 775 ml of water. calculate the following:
a) moles of k2so4
b)millimoles of k2so4
c)molarity of k2so4, k+, so4(2-)
d)ppm of k2so4
e)%(w/v) k2so4
f)pk+
g)pso4(2-)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2

Chemistry, 23.06.2019 00:30
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1

Chemistry, 23.06.2019 08:00
Suppose a pair of chemical compounds a and b can react in two different ways: a + b -> c reaction 1 gives product c. a + b -> d reaction 2 gives product d. the following facts are known about the two reactions: . reaction 1 is endothermic and reaction 2 is exothermic. if a reaction vessel is charged (filled) with a and b , then at first d is produced faster than c. use these facts to sketch a qualitative reaction energy diagram for both reactions. note: because these sketches are only qualitative, the energies don? t have to be exact. they only have to have the right relationship to each other. for example, if one energy is less than another, that fact should be clear in your sketch.
Answers: 3
You know the right answer?
a solution was prepared by dissolving 177 mg of potassium sulfate (k2so4, mw = 174.24 g/mol) in 775...
Questions


Social Studies, 23.11.2019 02:31








Computers and Technology, 23.11.2019 02:31





Computers and Technology, 23.11.2019 02:31





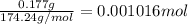
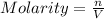
![[K_2SO_4]=\frac{0.001016 mol}{0.775 L}=0.001311 mol/L](/tpl/images/0275/4296/7b54d.png)
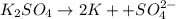
![[K^+]=2\times [K_2SO_4]=2\times 0.001311 mol/L=0.002622 mol/L](/tpl/images/0275/4296/87b7a.png)
![[SO_4^{2-}]=1\times [K_2SO_4]=1\times 0.001311 mol/L=0.001311 mol/L](/tpl/images/0275/4296/fd0e3.png)
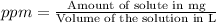
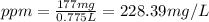
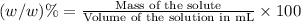
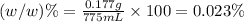
![pK^=-\log[K^+]](/tpl/images/0275/4296/a9ce4.png)
![pK^+=-\log[0.002622 M]=2.58](/tpl/images/0275/4296/0acce.png)
![pSO_4^{2-}=-\log[SO_4^{2-}]](/tpl/images/0275/4296/bd352.png)
![pK^+=-\log[0.001311 M]=2.88](/tpl/images/0275/4296/30697.png)


