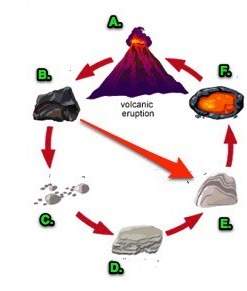
compute the theoretical yield of the product (in

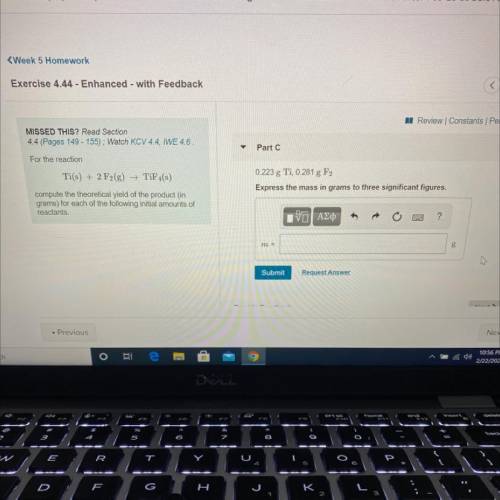
For the reaction
Ti(s) + 2 F2(g) →TiF4(s)
compute the theoretical yield of the product (in
grams) for each of the following initial amounts of
reactants.
0.223g Ti, 0.281g F2
Express your answer using three significant figures.
please help! will give brainliest.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Describe in detail the melting point behavior of the 80: 20 benzoic acid-mandelic acid mixture
Answers: 3

Chemistry, 21.06.2019 23:50
2points what is the job of a scientist? a. to answer ethical questions. b. to write laws based on his or her knowledge. c. to ask and answer scientific questions. d. to ignore facts that do not support his or her theory.
Answers: 1

Chemistry, 22.06.2019 03:00
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2
You know the right answer?
For the reaction
Ti(s) + 2 F2(g) →TiF4(s)
compute the theoretical yield of the product (in
compute the theoretical yield of the product (in
Questions



Social Studies, 05.10.2019 03:40





Mathematics, 05.10.2019 03:40

Mathematics, 05.10.2019 03:40

Biology, 05.10.2019 03:40

Mathematics, 05.10.2019 03:40

Mathematics, 05.10.2019 03:40


Mathematics, 05.10.2019 03:40

History, 05.10.2019 03:40

Computers and Technology, 05.10.2019 03:40

Social Studies, 05.10.2019 03:40

Biology, 05.10.2019 03:40

Biology, 05.10.2019 03:40