Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:30
What is the molecular formula of a hydrocarbon with m+ = 166? (write the formula with no subscripts, e.g. c4h10.) what is the sum of rings and double bonds in this compound?
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1


You know the right answer?
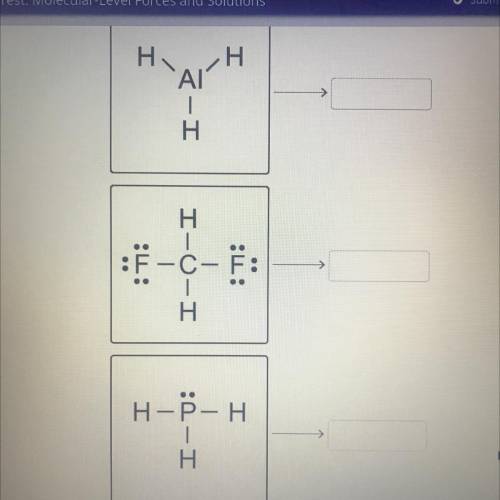
Drag the tiles to the correct boxes. Not all tiles will be used.
Match the molecular shapes to the...
Questions









Physics, 06.05.2020 22:11



Mathematics, 06.05.2020 22:11



Mathematics, 06.05.2020 22:11



Mathematics, 06.05.2020 22:11

Mathematics, 06.05.2020 22:11

Mathematics, 06.05.2020 22:11