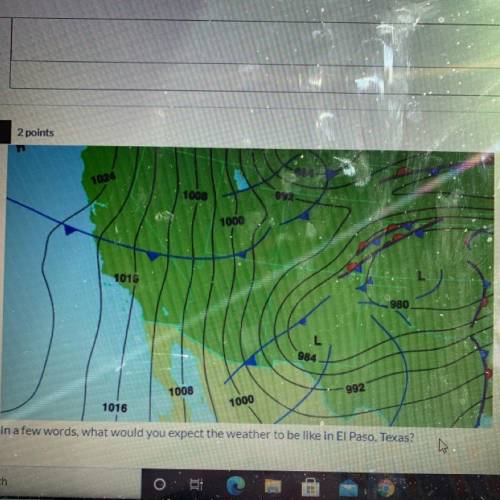
In a few words, what would you expect the weather to be like in El Paso, Texas?
...


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
Questions

Chemistry, 14.09.2020 04:01

Social Studies, 14.09.2020 04:01

History, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Biology, 14.09.2020 04:01

History, 14.09.2020 04:01

Physics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 04:01

Mathematics, 14.09.2020 05:01

Mathematics, 14.09.2020 05:01

Physics, 14.09.2020 05:01

Mathematics, 14.09.2020 05:01