
Chemistry, 24.02.2021 22:10 ftbluedevil
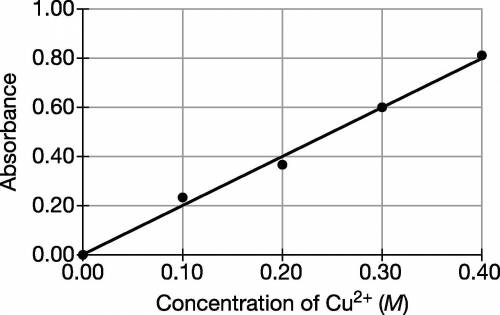
A student is given a sample of CuSO4(s) that contains a solid impurity that is soluble and colorless. The student wants to determine the amount of CuSO4 in the sample and decides to use a spectrophotometer. First, the student prepares a calibration graph by measuring the absorbances of CuSO4(aq) solutions of known concentrations. The graph is shown below. (a) The student dissolves the entire impure sample of CuSO4(s) in enough distilled water to make 100.mL of solution. Then the student measures the absorbance of the solution and observes that it is 0.30. Determine the concentration of CuSO4(aq) in the solution.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Sex cells from female reproductive organ? 1) mitosis 2) fertilization 3) zygote 4) eggs 5) meiosis 6) sperm
Answers: 2

Chemistry, 22.06.2019 01:30
There are main groups in the modern periodic table of elements
Answers: 1

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2
You know the right answer?
A student is given a sample of CuSO4(s) that contains a solid impurity that is soluble and colorless...
Questions

Mathematics, 12.03.2021 22:30

Mathematics, 12.03.2021 22:30



Mathematics, 12.03.2021 22:30

Mathematics, 12.03.2021 22:30

Mathematics, 12.03.2021 22:30

Mathematics, 12.03.2021 22:30



Mathematics, 12.03.2021 22:30

Mathematics, 12.03.2021 22:30



Mathematics, 12.03.2021 22:30



History, 12.03.2021 22:30




