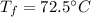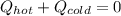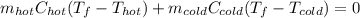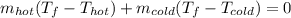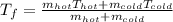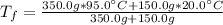Chemistry, 25.02.2021 08:10 rustalex6045
1. Mixing Water at Two Temperatures
a. One flask contains 150.0 g water at 20.0 °C. A second flask contains 350.0 g water at 95.0 °C. If the two water samples are mixed, what will the final temperature of the water be?
-Assume that the density of water it 1.000 g/cm^3
-The specific heat capacity for water is 4.184 J/g °C

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
The tilt of the earth's axis of rotation is responsible for the a) ocean's tides. b) size of the moon. c) brightness of stars. d) earth’s seasons.
Answers: 1

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1

Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3
You know the right answer?
1. Mixing Water at Two Temperatures
a. One flask contains 150.0 g water at 20.0 °C. A second flask...
Questions

Physics, 29.10.2020 01:20

Mathematics, 29.10.2020 01:20

History, 29.10.2020 01:20

Mathematics, 29.10.2020 01:20

English, 29.10.2020 01:20


Mathematics, 29.10.2020 01:20



Business, 29.10.2020 01:20

Mathematics, 29.10.2020 01:20

English, 29.10.2020 01:20

Advanced Placement (AP), 29.10.2020 01:20


History, 29.10.2020 01:20

Mathematics, 29.10.2020 01:20



English, 29.10.2020 01:20

Mathematics, 29.10.2020 01:20