
Chemistry, 26.02.2021 01:10 20stirltrer
WILL MARK BRAINLEST
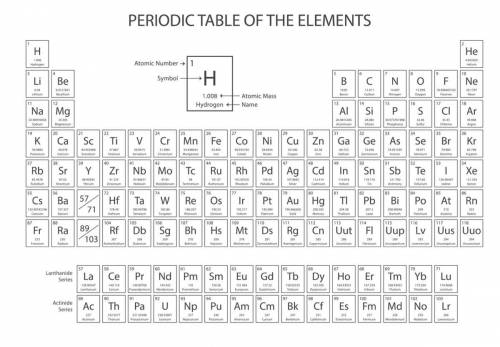
Look at the periodic table below. Which of the following lists of elements forms a group on the periodic table?
periodic table
Group of answer choices
Sc, Ti, V, Cr, Mn, Fe, Co, Cu, Ni, and Zn
He, Ne, Ar, Kr, Xe, and Rn
Li, Be, B, C,

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
If 34.2 grams of lithium react with excess water, how many liters of hydrogen gas can be produced at 299 kelvin and 1.21 atmospheres? 2 li (s) + 2 h2o (l) yields 2 lioh (aq) + h2 (g)
Answers: 3

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
WILL MARK BRAINLEST
Look at the periodic table below. Which of the following lists of elements form...
Questions

Mathematics, 27.07.2019 10:30

Chemistry, 27.07.2019 10:30

Mathematics, 27.07.2019 10:30



Chemistry, 27.07.2019 10:30

Mathematics, 27.07.2019 10:30


Mathematics, 27.07.2019 10:30

Chemistry, 27.07.2019 10:30

History, 27.07.2019 10:30

Business, 27.07.2019 10:30

History, 27.07.2019 10:30







Social Studies, 27.07.2019 10:30



