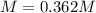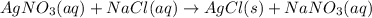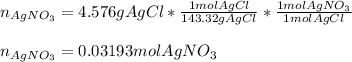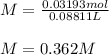Chemistry, 26.02.2021 14:00 deadman7628
In an experiment, a 88.11 mL sample of unknown silver nitrate solution was treated with 9.753 g of sodium chloride, resulting in 4.576 g of precipitate. Calculate the molarity of the silver nitrate solution

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
In an experiment, a 88.11 mL sample of unknown silver nitrate solution was treated with 9.753 g of s...
Questions

English, 21.01.2022 03:50

Mathematics, 21.01.2022 03:50



Mathematics, 21.01.2022 03:50


Mathematics, 21.01.2022 03:50






History, 21.01.2022 03:50

Business, 21.01.2022 03:50



Chemistry, 21.01.2022 03:50