
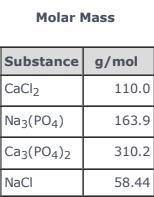
3CaCl2(aq) + 2Na3(PO4)(aq) → Ca3(PO4)2(s)+ 6NaCl(aq) Use the balanced equation and the Molar Mass table above to answer the following question. How much Ca3(PO4)2(s) could be produced in an industrial process if 55.00 g of CaCl2 in solution reacted completely with sufficient Na3(PO4)(aq)?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
You know the right answer?
3CaCl2(aq) + 2Na3(PO4)(aq) → Ca3(PO4)2(s)+ 6NaCl(aq) Use the balanced equation and the Molar Mass ta...
Questions




English, 04.06.2021 18:30

Mathematics, 04.06.2021 18:30

Biology, 04.06.2021 18:30

Physics, 04.06.2021 18:30


Mathematics, 04.06.2021 18:30

Mathematics, 04.06.2021 18:30

English, 04.06.2021 18:30


Mathematics, 04.06.2021 18:30





History, 04.06.2021 18:30





