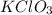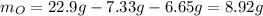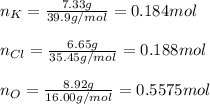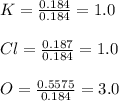Chemistry, 01.03.2021 08:50 noreenhussain
Help?
A 22.98 g sample of a compound contains 7.33 g of potassium, K, 6.65 g of chlorine, Cl, and oxygen, O. Calculate the empirical formula.
Insert subscripts as needed.
Empirical formula: KClO

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3


Chemistry, 23.06.2019 10:30
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 3.75 mol fe and 8.70 mol nio(oh) react?
Answers: 1

Chemistry, 23.06.2019 15:30
The amount of iron in ore can be quantitatively determined by titrating a solution of the unknown with a standard solution of dichromate, cr2o72−. the net ionic equation is 6fe2+(aq)+cr2o72−(aq)+14h+(aq)→6fe3+(aq)+2cr3+(aq)+7h2o(aq) part a the titration of 25.0 ml of an iron(ii) solution required 18.0 ml of a 0.230 m solution of dichromate to reach the equivalence point. what is the molarity of the iron(ii) solution?
Answers: 1
You know the right answer?
Help?
A 22.98 g sample of a compound contains 7.33 g of potassium, K, 6.65 g of chlorine, Cl, and o...
Questions

Mathematics, 25.02.2021 01:00

Mathematics, 25.02.2021 01:00



Biology, 25.02.2021 01:00

History, 25.02.2021 01:00







History, 25.02.2021 01:00

English, 25.02.2021 01:00

Mathematics, 25.02.2021 01:00

Mathematics, 25.02.2021 01:00

Mathematics, 25.02.2021 01:00

Mathematics, 25.02.2021 01:00

Mathematics, 25.02.2021 01:00

Social Studies, 25.02.2021 01:00