Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3
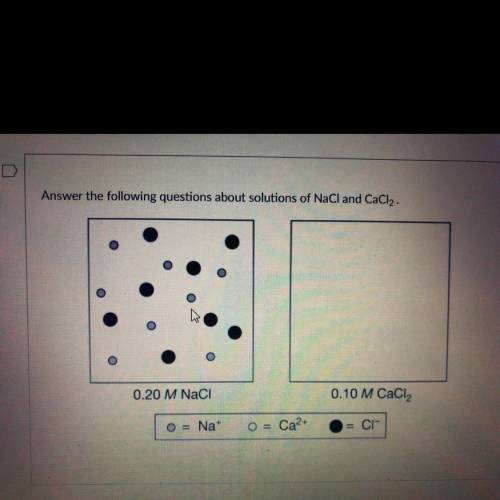
You know the right answer?
Ions in a certain volume of 0.20 M NaCl (aq) are represented in the box on the left. In the box abov...
Questions




Computers and Technology, 01.04.2021 18:40

English, 01.04.2021 18:40






Computers and Technology, 01.04.2021 18:40


Mathematics, 01.04.2021 18:40



Mathematics, 01.04.2021 18:40

History, 01.04.2021 18:40

English, 01.04.2021 18:40