
Chemistry, 03.03.2021 01:40 trentonmccary2096
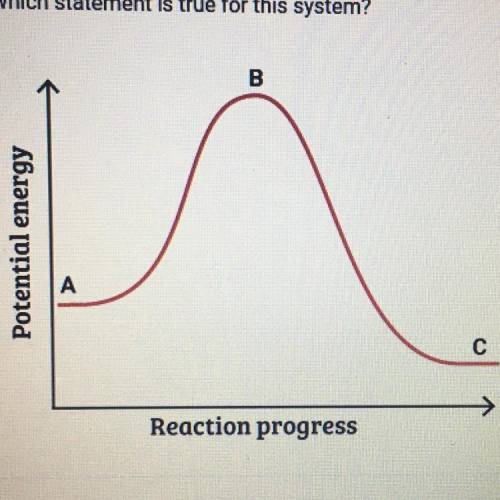
This graph shows how the potential energy of a reaction system changes
over time. Which statement is true for this system
A. The potential energy of the reactants is greater than the potential
energy of the products.
B. The height of the curve at point A represents the activation energy.
C. The height of the curve at point B represents the activation energy.
D. The potential energy of the products is greater than the potential
energy of the reactants.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1


Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
This graph shows how the potential energy of a reaction system changes
over time. Which statement i...
Questions


Physics, 05.11.2020 22:30

Spanish, 05.11.2020 22:40

Mathematics, 05.11.2020 22:40

History, 05.11.2020 22:40

Mathematics, 05.11.2020 22:40



Geography, 05.11.2020 22:40

English, 05.11.2020 22:40

Social Studies, 05.11.2020 22:40

History, 05.11.2020 22:40

Chemistry, 05.11.2020 22:40


Arts, 05.11.2020 22:40

History, 05.11.2020 22:40


Mathematics, 05.11.2020 22:40




