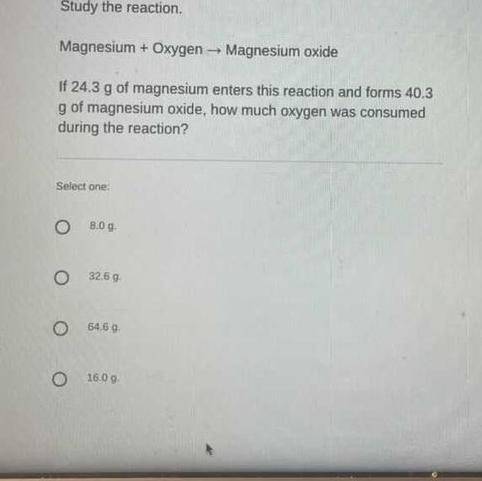
Help! Please and thank you
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1


Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Questions

Chemistry, 28.01.2021 14:00

English, 28.01.2021 14:00


English, 28.01.2021 14:00



Mathematics, 28.01.2021 14:00

English, 28.01.2021 14:00

Mathematics, 28.01.2021 14:00



Mathematics, 28.01.2021 14:00


Mathematics, 28.01.2021 14:00






Mathematics, 28.01.2021 14:00