
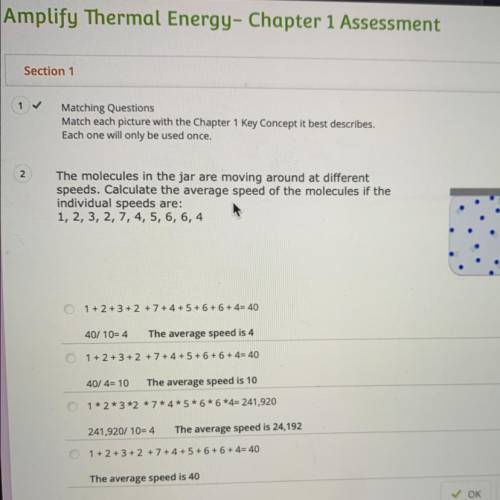
The molecules in the jar are moving around at different
speeds. Calculate the average speed of the molecules if the
individual speeds are:
1, 2, 3, 2, 7, 4, 5, 6, 6, 4
1 + 2 + 3+2 +7+4+ 5+ 6 + 6 + 4= 40
40/ 10=4
The average speed is 4
1 + 2 + 3 + 2 + 7 + 4 + 5+ 6 + 6 + 4= 40
40/4= 10
The average speed is 10
1*2*3*2*7*4*5*6*6*4= 241,920
241,920/ 10=4
The average speed is 24,192
1 + 2 + 3 + 2 + 7 + 4 + 5 + 6 + 6 + 4= 40
The average speed is 40

Answers: 3
Another question on Chemistry


Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 06:00
•what conclusions can you make about the relationship between the volume of a gas and its temperature? • what conclusions can you make about the relationship between the volume of a gas and its pressure? • what possible variables have you not accounted for? as you did the procedures, is it possible that the atmospheric pressure may have changed? if it did change over the course of your experiment, then how would your results have been affected?
Answers: 3

Chemistry, 23.06.2019 06:30
The molar mass of cu is 63.55 g/mol. the number of grams of cu produced in this reaction is
Answers: 3
You know the right answer?
The molecules in the jar are moving around at different
speeds. Calculate the average speed of the...
Questions

English, 05.02.2020 06:44



Social Studies, 05.02.2020 06:44


Spanish, 05.02.2020 06:44

Mathematics, 05.02.2020 06:44


English, 05.02.2020 06:45

Mathematics, 05.02.2020 06:45

Computers and Technology, 05.02.2020 06:45

Chemistry, 05.02.2020 06:45

Health, 05.02.2020 06:45


History, 05.02.2020 06:45



Mathematics, 05.02.2020 06:45

Mathematics, 05.02.2020 06:45



