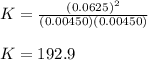Chemistry, 05.03.2021 14:00 hoopstarw4438
At equilibrium at 2500K, [HCl]=0.0625M and [H2]=[Cl2]=0.00450M for the reaction H2+Cl2 ⇌ HCl.
a. Determine the balanced equation and write the equilibrium expression
b. Determine the K eq
c. Will this process favor the reactants or products at equilibrium?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
In the analysis of hair and fiber samples, which does a compound comparison microscope allow for that a conventional compound microscope does not? a. simultaneous observation b. polarization c. fluorescence d. higher magnification
Answers: 2

Chemistry, 22.06.2019 00:40
Which is a difference between molecular compounds and ionic compounds? select the correct answer below: question 5 options: molecular compounds typically form between a metal and a nonmetal, while ionic compounds typically form between nonmetals. molecular compounds result from the transfer of electrons between atoms to form ions, while ionic compounds result from the sharing of electrons between neutral atoms. molecular compounds are formed of discrete, neutral molecules, while ionic compounds are formed of large repeating arrays of opposite charges. molecular compounds have high melting points and high boiling points, while ionic
Answers: 3

Chemistry, 22.06.2019 05:20
Why does the sun appear to be the brightest star in the sky? a- its apparent brightness is much greater than other stars. b- it burns more gas, making it brighter than any other star. c- it is the largest star in the galaxy, so it is the brightest star. d- its relative distance to earth is closer than the other stars.
Answers: 1

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2
You know the right answer?
At equilibrium at 2500K, [HCl]=0.0625M and [H2]=[Cl2]=0.00450M for the reaction H2+Cl2 ⇌ HCl.
a. De...
Questions

Mathematics, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00


Biology, 05.07.2019 02:00


History, 05.07.2019 02:00

English, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00

English, 05.07.2019 02:00

Business, 05.07.2019 02:00

Social Studies, 05.07.2019 02:00

Social Studies, 05.07.2019 02:00


History, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00


History, 05.07.2019 02:00

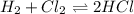
![K=\frac{[HCl]^2}{[H_2][Cl_2]}](/tpl/images/1171/4277/48ea9.png)