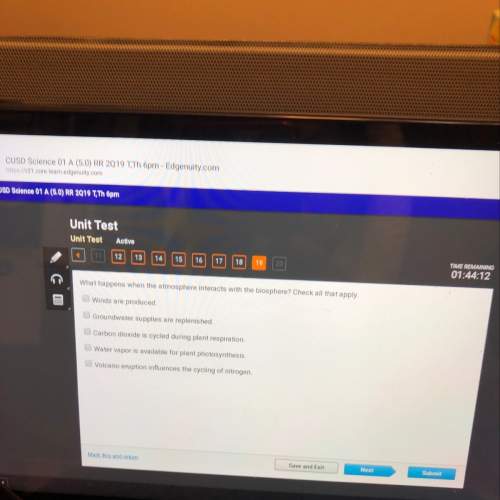
Given the empirical formula C3H4O2, what is the molecular formula that has a mass of
288.6g?...

Chemistry, 08.03.2021 17:20 jackie6852
Given the empirical formula C3H4O2, what is the molecular formula that has a mass of
288.6g?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of the dna typing techniques do you think you would choose if you had to analyze a dna sample? why?
Answers: 1

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3


Chemistry, 23.06.2019 00:30
Fred is studying a substance that is made out of only one element. this means that
Answers: 1
You know the right answer?
Questions


Mathematics, 06.02.2021 21:10

Biology, 06.02.2021 21:10

Physics, 06.02.2021 21:10



Business, 06.02.2021 21:10

Business, 06.02.2021 21:10

Mathematics, 06.02.2021 21:10



Physics, 06.02.2021 21:10


Chemistry, 06.02.2021 21:10


Biology, 06.02.2021 21:10


Mathematics, 06.02.2021 21:10