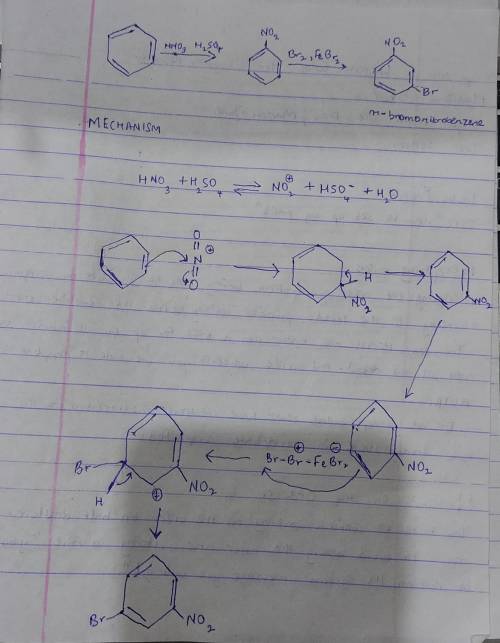
Chemistry, 08.03.2021 19:10 groovyghoulie5630
Select the reagents you would use to synthesize the compounds below from benzene. Use the minimum number of steps.
a. Br2, FeBr3
b. CH3COCl, AlCl3
c. Cl2, FeCl3
d. CH3Cl, AlCl3

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1


Chemistry, 22.06.2019 13:30
Some animals that try to adapt to climate changes eventually die due to starvation, as climate change alters the web.
Answers: 2

Chemistry, 22.06.2019 18:40
What is one real world example of a colligative property?
Answers: 2
You know the right answer?
Select the reagents you would use to synthesize the compounds below from benzene. Use the minimum nu...
Questions









History, 20.03.2020 06:11

Mathematics, 20.03.2020 06:11



Mathematics, 20.03.2020 06:11



Mathematics, 20.03.2020 06:12