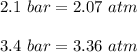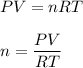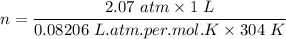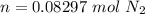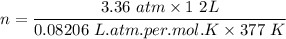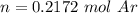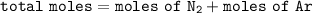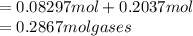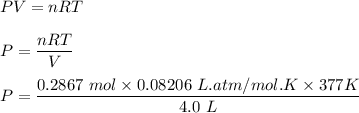Chemistry, 12.03.2021 15:30 Yasminl52899
One liter of N (g) at 2.1 bar and two liters of Ar(g) at 3.4 bar are mixed in a 4.0-L 2 flask to form an ideal-gas mixture. Calculate the value of the final pressure of the mixture if the initial and final temperature of the gases are the same. Repeat this calculation if the initial temperatures of the N (g) and Ar(g) are 304 K and 402 K, respectively, and the final 2 temperature of the mixture is 377 K. (Assume ideal-gas behavior.)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 04:50
Write the overall equation for the reaction for lithium battery
Answers: 2

Chemistry, 22.06.2019 06:30
The minerals found in bones are deposited by living cells called
Answers: 1

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2
You know the right answer?
One liter of N (g) at 2.1 bar and two liters of Ar(g) at 3.4 bar are mixed in a 4.0-L 2 flask to for...
Questions

Biology, 02.08.2019 05:00

History, 02.08.2019 05:00





Computers and Technology, 02.08.2019 05:00

Biology, 02.08.2019 05:00


Health, 02.08.2019 05:00



Health, 02.08.2019 05:00



Mathematics, 02.08.2019 05:00



Mathematics, 02.08.2019 05:00

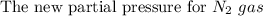
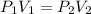
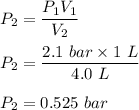
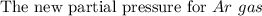
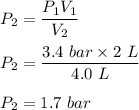
![Total pressure= P [N_2] + P[Ar] \ \\ \\ . \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ = (0.525 + 1.7)Bar \\ \\ . \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ = 2.225 \ Bar](/tpl/images/1191/0157/39a5e.png)