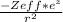Chemistry, 13.03.2021 05:50 HeyItsCookie9605
Describe the critical property ofelectrons in 2s versus 2p orbitals that causes the radial distribution functions of these orbitals to have different shapes about the nucleus such that 2s electrons effectively penetrate closer to the nucleus than do electrons in 2p orbitals. Then, write an expression for the potential energy function describing this effect.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:50
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1
You know the right answer?
Describe the critical property ofelectrons in 2s versus 2p orbitals that causes the radial distribut...
Questions





History, 04.01.2020 00:31




Mathematics, 04.01.2020 00:31

Geography, 04.01.2020 00:31



English, 04.01.2020 00:31


English, 04.01.2020 00:31

English, 04.01.2020 00:31

Mathematics, 04.01.2020 00:31


English, 04.01.2020 00:31