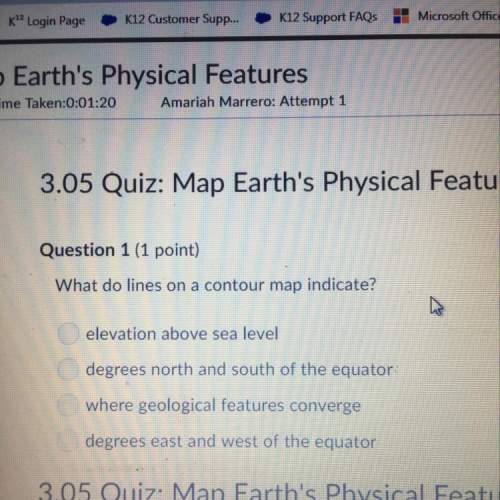
Chemistry, 18.03.2021 01:00 raytaygirl
11) A fixed amount of gas at 25.0 °C occupies a volume of 8.66 L when the pressure is 629 torr. Use
Charles's law to calculate the volume (L) the gas will occupy when the temperature is increased to
112 °C while maintaining the pressure at 629 torr.
A) 1.93
B) 38.8
C) 11.2
D) 9.26
E) 6.70

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
You know the right answer?
11) A fixed amount of gas at 25.0 °C occupies a volume of 8.66 L when the pressure is 629 torr. Use...
Questions

Mathematics, 01.07.2019 07:20

Mathematics, 01.07.2019 07:20

English, 01.07.2019 07:20


Biology, 01.07.2019 07:20

Biology, 01.07.2019 07:20

History, 01.07.2019 07:20


Business, 01.07.2019 07:20

Social Studies, 01.07.2019 07:20


Chemistry, 01.07.2019 07:20




Mathematics, 01.07.2019 07:20



Mathematics, 01.07.2019 07:20