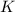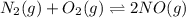Chemistry, 18.03.2021 01:10 alexandrecarmene
The equilibrium-constant expression is used to describe the concentration of reactants and products for a reaction in dynamic equilibrium. For ideal gases and ideal solutions in homogeneous equilibria, where all reactants and products are in the same phase, the extent to which a particular chemical reaction proceeds to products is given by the equilibrium equation.
aA+bB⇌cC+dD , K=[C]c[D]d[A]a[B]b
where K is the equilibrium constant and the right-hand side of the equation is known as the equilibrium-constant expression.
The concentration of each product raised to its coefficient is divided by the concentration of each reagent raised to its coefficient according the the balanced chemical equation. Therefore, the higher the concentration of products, the larger the value of K will be.
Required:
Identify the proper form of the equilibrium-constant expression for the equation.
N2(g)+O2(g)⇌2NO(g)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Earth's axis of rotation is tilted at an angle of 23.5 degrees. what is one change you would see on earth if its axis was not tilted?
Answers: 3

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2
You know the right answer?
The equilibrium-constant expression is used to describe the concentration of reactants and products...
Questions



Physics, 22.07.2019 15:50

Mathematics, 22.07.2019 15:50



Mathematics, 22.07.2019 15:50


Mathematics, 22.07.2019 15:50




Mathematics, 22.07.2019 15:50



Social Studies, 22.07.2019 15:50




![K=\frac{[NO]^2}{[N_2]^1[O_2]^1}](/tpl/images/1195/8832/df6dc.png)