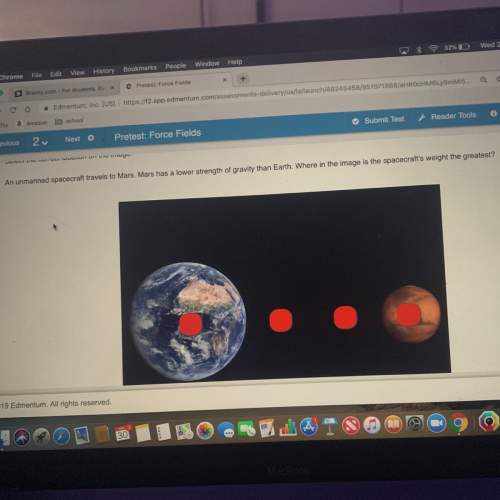
Chemistry, 18.03.2021 02:50 rowdycar313p0ao5k
The specific heat of water is 4.18 J/g•°C. How much heat does 150.0 g of water release when it cools from 92.0°C to 40.0°C? You must show all of your work to receive credit. Your answer must include the correct unit. Q=mcTriangleT

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2


Chemistry, 23.06.2019 06:40
A250 g sample of water with an initial temperatureof 98.8 closes 6500 joules of heat. what is the finaltemperature of the water?
Answers: 1
You know the right answer?
The specific heat of water is 4.18 J/g•°C. How much heat does 150.0 g of water release when it cools...
Questions


Mathematics, 29.08.2021 20:20


Social Studies, 29.08.2021 20:20

Social Studies, 29.08.2021 20:20

Engineering, 29.08.2021 20:20

Mathematics, 29.08.2021 20:20





Computers and Technology, 29.08.2021 20:20

Mathematics, 29.08.2021 20:20

Biology, 29.08.2021 20:20


Mathematics, 29.08.2021 20:20

Mathematics, 29.08.2021 20:20

Physics, 29.08.2021 20:20

Mathematics, 29.08.2021 20:20