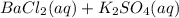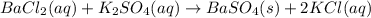Chemistry, 18.03.2021 02:50 Gimagg5549
What are the products from the following double - replacement reaction ? BaCl 2(aq) +K 2 SO 4(aq) a . BaSO 3 and KCI b. and KClO 4 c. BaSO 3 KClO 4 d. BaSO 4 and KClO 4 e . BaSO 4 and KCI

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:20
Amixture of gaseous sulfur dioxide and oxygen are added to a reaction vessel and heated to 1000 k where they react to form so3(g). if the vessel contains 0.669 atm so2(g), 0.395 atm o2(g), and 0.0851 atm so3(g) after the system has reached equilibrium, what is the equilibrium constant kp for the reaction: 2 so2(g) o2(g) ⇌ 2 so3(g)
Answers: 3

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 15:10
The ozone molecule o3 has a permanent dipole moment of 1.8×10−30 cm. although the molecule is very slightly bent-which is why it has a dipole moment-it can be modeled as a uniform rod of length 2.5×10−10 m with the dipole moment perpendicular to the axis of the rod. suppose an ozone molecule is in a 8000 n/c uniform electric field. in equilibrium, the dipole moment is aligned with the electric field. but if the molecule is rotated by a small angle and released, it will oscillate back and forth in simple harmonic motion.what is the frequency f of oscillation?
Answers: 2

Chemistry, 23.06.2019 00:00
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1
You know the right answer?
What are the products from the following double - replacement reaction ? BaCl 2(aq) +K 2 SO 4(aq) a...
Questions


French, 19.03.2021 20:30

Chemistry, 19.03.2021 20:30


Mathematics, 19.03.2021 20:30




Social Studies, 19.03.2021 20:30



Computers and Technology, 19.03.2021 20:30

Physics, 19.03.2021 20:30



Mathematics, 19.03.2021 20:30


Mathematics, 19.03.2021 20:30


Mathematics, 19.03.2021 20:30