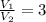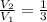Chemistry, 18.03.2021 14:00 nelsoneligwe7
1. The pressure of a gas is 100.0 kPa and its volume is 500.0 ml. If the volume increases to 1,000.0 ml, what is the new pressure of the gas?
2. If a gas at 25.0 °C occupies 3.60 liters at a pressure of 10 kPa, what will be its volume at a pressure of 25 kPa?
3. When the pressure on a gas increases three times, by how much will the volume increase or decrease?
4. Boyle's Law deals what quantities?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1

Chemistry, 22.06.2019 12:00
Hey guys so i need to know what is _nh3+> nh4oh ~chemistry~
Answers: 1

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

You know the right answer?
1. The pressure of a gas is 100.0 kPa and its volume is 500.0 ml. If the volume increases to 1,000.0...
Questions


Mathematics, 07.10.2021 14:00

Mathematics, 07.10.2021 14:00

Social Studies, 07.10.2021 14:00

Business, 07.10.2021 14:00

Health, 07.10.2021 14:00

Computers and Technology, 07.10.2021 14:00


Mathematics, 07.10.2021 14:00

Computers and Technology, 07.10.2021 14:00

Social Studies, 07.10.2021 14:00

Social Studies, 07.10.2021 14:00


Mathematics, 07.10.2021 14:00

Computers and Technology, 07.10.2021 14:00


Physics, 07.10.2021 14:00


Computers and Technology, 07.10.2021 14:00

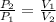 (1)
(1)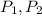 - Initial and final pressure, measured in kPa.
- Initial and final pressure, measured in kPa.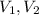 - Initial and final pressure, measured in mililiters.
- Initial and final pressure, measured in mililiters.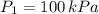 ,
, 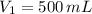 and
and 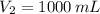 , then the new pressure of the gas is:
, then the new pressure of the gas is: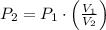
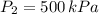
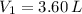 ,
, 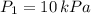 and
and 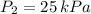 then the new volume of the gas is:
then the new volume of the gas is: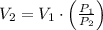
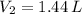
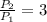 , then the volume ratio is:
, then the volume ratio is: