Chemistry, 18.03.2021 21:40 alyssalefeber
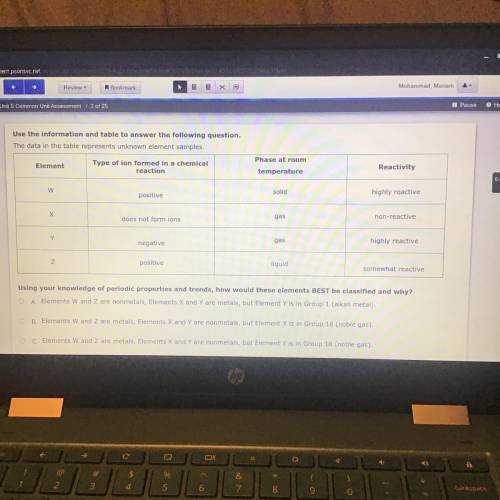
Use the information and table to answer the following question.
The data in the table represents unknown element samples.
Using your knowledge of periodic properties and trends, how would these elements BEST be classified and why?
O A. Elements W and Z are nonmetals, Elements X and Y are metals, but Element Y is in Group 1 (alkali metal).
O B. Elements W and Z are metals, Elements X and Y are nonmetals, but Element X is in Group 18 (noble gas).
O C. Elements W and Z are metals, Elements X and Y are nonmetals, but Element Y is in Group 18 (noble gas).
D.) Elements W and Z nonmetals, what element W is in group 17(halogen) Element X and Y are metals

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Balance this equation: n2 + h2 > nh3, write the following molar ratios: n2 / n2 / nh3 h2 /
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

You know the right answer?
Use the information and table to answer the following question.
The data in the table represents un...
Questions



Mathematics, 02.05.2021 06:40


Mathematics, 02.05.2021 06:40

Social Studies, 02.05.2021 06:40


English, 02.05.2021 06:40



Social Studies, 02.05.2021 06:40


Social Studies, 02.05.2021 06:40

English, 02.05.2021 06:40





Mathematics, 02.05.2021 06:40

Mathematics, 02.05.2021 06:40