
Chemistry, 18.03.2021 23:40 larueeee25
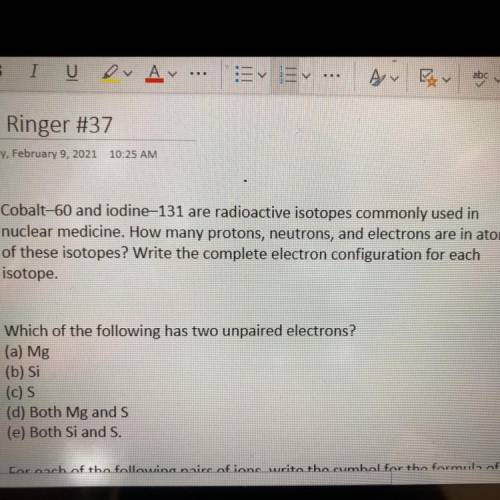
Cobalt-60 and iodine 131 are radioactive isotopes commonly used in
nuclear medicine. How many protons, neutrons, and electrons are in atoms
of these isotopes? Write the complete electron configuration for each
isotope.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Embryos of different species look very similar, which shows that the organisms share a ancestor.
Answers: 1

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2
You know the right answer?
Cobalt-60 and iodine 131 are radioactive isotopes commonly used in
nuclear medicine. How many proto...
Questions


Mathematics, 15.12.2021 04:40

Mathematics, 15.12.2021 04:40





Mathematics, 15.12.2021 04:40

Business, 15.12.2021 04:40




English, 15.12.2021 04:40

History, 15.12.2021 04:40



Business, 15.12.2021 04:50

English, 15.12.2021 04:50

Mathematics, 15.12.2021 04:50

Biology, 15.12.2021 04:50



