Chemistry, 19.03.2021 05:50 taufajane3887
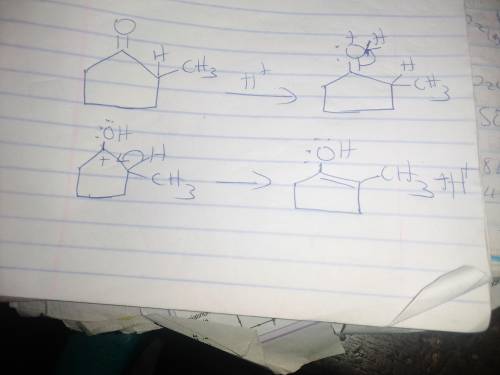
When optically active (S)-2-methylcyclopentanone is treated with an acid (H3O ), the compound loses its optical activity. Explain this observation and draw a mechanism that shows how racemization occurs. For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly in your products. Do not use abbreviations such as Me or Ph.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is lincoln's purpose in writing this speech? question 1 options: to stress the difficulties of war to honor those who died in the war to call for an end to the war to call the country to join a new war
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
You know the right answer?
When optically active (S)-2-methylcyclopentanone is treated with an acid (H3O ), the compound loses...
Questions




Mathematics, 06.05.2020 06:41

Mathematics, 06.05.2020 06:41


Mathematics, 06.05.2020 06:41




Chemistry, 06.05.2020 06:41



Medicine, 06.05.2020 06:41





Mathematics, 06.05.2020 06:41

History, 06.05.2020 06:41